A recent study demonstrates a point-of-care electrochemical biosensing workflow to quantify an Alzheimer’s-relevant marker on neuron-origin exosomes isolated from plasma. The assay is implemented in a disposable, magneto-actuated cartridge and the electrochemical signal is acquired using BioEclosion’s portable device.
Early diagnosis of Alzheimer’s disease remains one of the major challenges in healthcare, largely because many current diagnostic pathways are expensive, invasive, or difficult to deploy at scale. This is why blood-based biomarkers are receiving increasing attention—especially those that can provide brain-related molecular information from minimally invasive samples.
Among the most promising candidates are plasma brain-derived exosomes (BDEs): nano-sized extracellular vesicles that can carry disease-relevant cargo and enable a “liquid biopsy” approach. However, translating exosome biomarkers into practical diagnostics requires two things at once: selective isolation of the relevant vesicle subpopulation and a sensitive readout compatible with decentralized testing.
In a newly published study, researchers propose a dual-biomarker immunoassay that selectively isolates neuron-origin exosomes using neuroligin-3 (NLGN3) as the capture marker, followed by detection of β-secretase 1 (BACE-1), a key enzyme linked to Alzheimer’s pathology. This combination (NLGN3 for capture and BACE-1 for detection) is presented as a first-of-its-kind exosomal biomarker strategy aimed at improving biological specificity for neurodegenerative diagnostics.
From BioEclosion’s perspective, a key highlight is the integration of this assay into a portable electrochemical biosensing format. The study evaluates multiple detection approaches and advances the electrochemical readout as a strong candidate for point-of-care use—combining sensitivity with compact instrumentation and disposable electrodes.
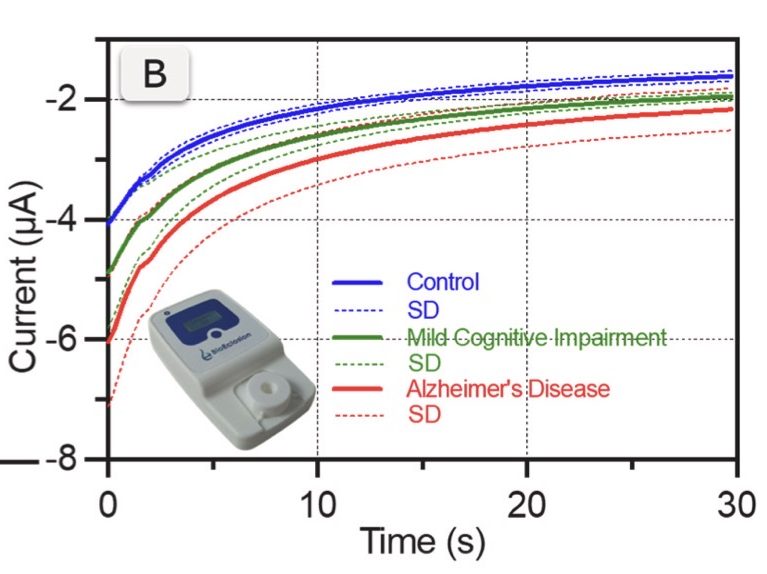
Crucially, the study uses BioEclosion’s point-of-care electrochemical device for signal acquisition. Electrochemical measurements are performed with a handheld potentiostat developed by BioEclosion, together with a single-use, magneto-actuated microfluidic cartridge integrating screen-printed electrodes. This cartridge-based approach supports practical workflows by combining magnetic handling (to simplify separation and washing) with a robust amperometric readout suitable for decentralized environments.
To explore clinical relevance, the platform is applied to plasma exosomes from pooled cohorts of healthy controls, mild cognitive impairment (MCI), and Alzheimer’s disease (AD) patients. The electrochemical response shows an increasing trend across groups (control → MCI → AD), supporting the potential of exosome-based biosensing for early-stage differentiation and patient stratification. As expected for a proof-of-concept, the authors note that larger cohorts and individual-level validation will be important next steps.
Overall, this work illustrates a clear direction for scalable, minimally invasive neurodegenerative screening: selective exosome capture, magnetic actuation for simplified handling, and portable electrochemical detection. At BioEclosion, we see this as a strong example of how magneto-actuated cartridges and portable electrochemical instrumentation can accelerate the translation of advanced biomarker assays into realistic IVD and point-of-care workflows.
Reference
Electrochemical biosensor for early Alzheimer’s detection and patient risk stratification using plasma exosomes. Biosensors and Bioelectronics, 292 (2026) 118061. DOI: 10.1016/j.bios.2025.118061